

Silicate liquids of various compositions are the predominant products of melting in the Earth's crust and upper mantle. Rare examples of non-silicate melts (e.g., parental liquids of carbonatites and sulfide ores) are mostly restricted to evolved residual systems. There are good reasons to believe that they can be derived from silicate magmas by liquid immiscibility. In synthetic silicate systems liquid immiscibility is also common in the presence of F, Cl and B2O3, but terrestrial abundances of these elements are too low for any significant phase separation in natural melts. Although cases of liquid unmixing in nature are limited, the phenomenon has very important implications because it represents the most vivid manifestation of non-ideal mixing. Experimental studies of element partitioning between different pairs of immiscible liquids provide an insight into thermodynamics and speciation of inorganic liquids and enable better understanding of energetics and relative stability of chemical bonds.
Silicate - fluoride liquid immiscibility has been studied in the six-component
SiO2-Al2O3-MgO-CaO-K2O-F synthetic
system. Reactant mixtures were prepared from K aluminosilicate glasses
and reagent-grade CaF2 and MgF2 that cover a broad
range of K-Al and K-Si mole ratios: from peralkaline to peraluminous (2>K/Al>0.5)
and from SiO2-depleted to SiO2-oversaturated (0.5<(K+Al)/Si<4)
compositions. Nine identical starting compositions were used in two series
of runs at atmospheric pressure and temperatures of 1150-1400oC:
rapid quench runs were carried out in a vertical tube furnace at the Lamont-Doherty
Earth Observatory (LDEO, Palisades, N.Y.), and centrifuge separation in
the rotating furnace at the BGI. Due to the effects of centrifuge separation
(at 1000 g acceleration) and slower quench (2-3 minutes vs. 1-2 seconds
at the LDEO), the two sets of run products differ in texture. However,
the compositions of the immiscible liquids are very similar, suggesting
the attainment of chemical equilibrium. In the products of the rapid-quench
runs, the fluoride immiscible liquid formed spherical globules of variable
size (20-500 microns) randomly distributed in the silicate glass, whereas
centrifuge separation resulted in two distinct layers of immiscible liquids
with some small scattered globules in the silicate layer produced on quenching
(Fig. 3.2-6). Both sets of run products were analysed by electron microprobe
at the
 |
|
|
American Museum of Natural History, New York. Fluoride layers of the centrifuge runs, where the liquid crystallized into coarse-grained quench crystal aggregates, posed a significant analytical difficulty. In order to get representative average bulk compositions the layers were analyzed by beam scanning over areas that were large relative to the size of the quench crystals.
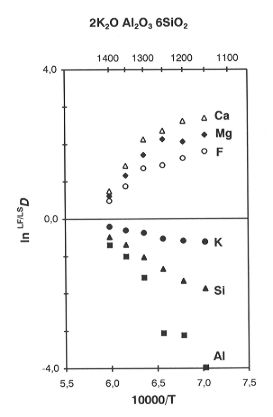
All of the runs fell into a two-liquid, above-liquidus region. At temperatures between 1350-1400oC some silica-undersaturated compositions reached the critical point, as revealed by unusual quench textures and convergent compositions of the liquids (Fig. 3.2-7). In general, Ca and Mg strongly partition to the fluoride liquid (LF), while Al and K are more compatible with the silicate melt (LS). The SiO2-saturated compositions appear to be far below the critical temperatures and the LF/LS partition coefficients (LF/LSD) do not change much within the temperature interval of our experiments.
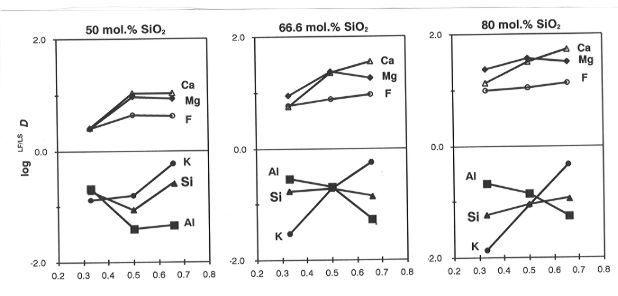
Partition coefficients (LF/LSD) depend strongly on the bulk
SiO2 content and K/Al mole ratio in the reactant mixtures. The
variations are summarized in Fig. 3.2-8, which presents three
 |
|
|
 |
|
|
isothermal (1250oC) joins of variable K/(K+Al) at three different levels of SiO2 saturation. On all the joins, LF/LSD for K and Al show strong and systematic variations in opposite directions: in peraluminous compositions LF/LSD Al > LF/LSD Na, while in peralkaline compositions the situation is reversed. Partitioning of Ca and Mg is also affected by the bulk K/(K+Al): Ca is much more compatible with peraluminous liquids than with peralkaline ones. These relationships imply that the network of tetrahedrally coordinated Al and Si is not destroyed or depolymerized in the presence of abundant F- anions, which form complexes with network-modifying cations. Phase separation is a consequence of the limited structural compatiblity of fluoride complexes with the polymerized alumosilicate network. The values of LF/LSD depend on the relative stability of fluoride complexes and chemical bonds of different network-modifiers with the alumosilicate network. As a typical amphoteric element Al plays a dual role in the silicate liquid: it is a 4-coordinated network-former and a 6-coordinated network-modifier at the same time and the proportions of the two coordination states depend on K/Al and (to lesser extent) Ca/Al values in the bulk compositions. Variations of LF/LSD with bulk composition result from a subtle balance between these structural and chemical interactions.
Ionic fluoride liquid appears to be closer to an ideal mixture than a polymerized and strongly non-ideal silicate melt. Quantitative data on element partitioning between the conjugate liquids may help to put constraints on concentration-activity relationships in F-bearing silicate liquids at variable temperatures for a broad range of bulk compositions.
The basic composition chosen for the study of silicate-borate liquid immiscibility is a three-component synthetic mixture CaO-B2O3-2SiO2 - a stoichiometry that corresponds to that of natural danburite. It lies in the middle of two-liquid field of the CaO - B2O3 - SiO2 system. In order to study the effect of different cations on phase separation several intermediate compositions have been prepared along the CaB2Si2O8 - NaBSi3O8 and CaB2Si2O8 - CaAl2Si2O8 joins. Some compositions were also doped with 5 wt% MgO. Experiments are carried out at 1 atm and 1050-1300oC. Due to the high viscosity of the liquids, full phase separation was achieved only at longer run durations (3 hours) and accelerations of 1000 g.
Run products were analysed by electron microprobe. Preliminary data on the compositions of immiscible liquids allow us to draw some general conclusions and point out directions for further studies:
1. Although boron can form triangular units similar to the CO32- group, element partitioning in silicate-borate systems is very different from that in the silicate carbonate systems. The distribution of network-forming elements (Si, B and Al) is much less variable and (contrary to silicate-carbonate systems) Al concentrates in the B-enriched conjugate liquid (LB). Network modifiers (Mg, Ca and Na) are strongly concentrated in LB. In general the LB/LS partition coefficients for network-modifying elements decrease in the order: Mg>Ca>Na and strongly depend on bulk composition.
2. The effect of B-Al substitution on phase separation in our basic CaO - B2O3 - 2SiO2 composition is much more pronounced than that of the Ca-Na substitution. The width of the miscibility gap rapidly decreases along the CaB2Si2O8 - CaAl2Si2O8 join and compositions with Al/B mole values > 0.54 do not show any macroscopic signs of phase separation. Compositions on the CaB2Si2O8 - NaAlSi3O8 join with Na/Ca mole values up to 1 show either stable or metastable liquid immiscibility.
3. In some Na-bearing compositions there are indications for a complex solvus shape and non-monotonous temperature dependence of element partitioning. Such relationships were observed between immiscible organic liquids, but have never been found in inorganic systems. Further chemical analyses of run products will provide a test of this phenomenon.

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page