

In the lower mantle, silicate perovskite is believed to be the dominant mineral. Recent experimental investigations (see Lauterbach et al., this report, and Annual Report 1997) have shown that the properties of these perovskites are strongly influenced by the incorporation of minor elements such as Fe and Al. This incorporation can result in the formation of oxygen-deficient perovskites and is expected to have strong influence on properties such as crystal structure and phase relations, electrical conductivity, diffusivity, incorporation of OH, etc. Detailed investigations of these effects in silicate perovskites are difficult because of the extreme conditions required for their synthesis (above 23 GPa for MgSiO3 perovskite), the instability of these phases in ex-situ investigations, and the expected low concentration of defects. We have therefore selected a more simple system to investigate some of these variables in the perovskite structure.
In the first stage of this project we are studying the phase transformations in the system CaTiO3-CaFeO2.5 as a function of temperature and composition. Members of this series, with general formula CaTi1-xFexO3-x/2, are obtained by chemical substitution of Ti4+ by Fe3+, which results in the creation of oxygen vacancies due to the different oxidation state of the cations involved. At high Fe contents (x=0.5) it is known that the vacancies are ordered, giving rise to the formation of layers of Fe tetrahedra. In these ordered structures with discrete compositions, the ratio of octahedral to tetrahedral layers decreases with Fe content, reaching 1 in the CaFeO2.5 end member (the mineral brownmillerite). The samples studied in this project (0 <x <0.5) were synthesised from mixtures of Fe2O3, TiO2 and CaCO3, decarbonated at 1050°C during 24 hours and sintered at 1350°C in air for 4 days. Then, annealing temperatures from 900°C to 1400°C and CO/CO2 mixtures at logO2 corresponding to IW+1 were employed in order to reduce the small amounts of Fe4+ to Fe3+. Finally, the samples were drop quenched and studied by ex-situ X-ray diffraction (XRD), transmission electron microscopy (TEM) and Mössbauer spectroscopy.
The phase diagram as a function of temperature and composition obtained
so far (Fig. 3.3-1)
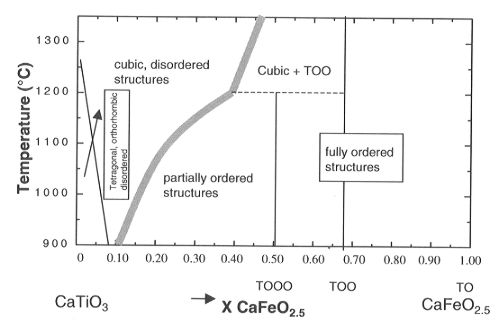 |
|
Fig. 3.3-1: Phase diagram of the solid solution CaTiO3-CaFeO2.5
as a function of temperature and composition. Symbols TO, TOO and TOOO
indicate the sequence of tetrahedral (T) and octahedral (O) layers in the
ordered structures.
|
shows the existence of two different types of phase transitions. At
Fe contents up to 40% CaFeO2.5 and high temperatures a cubic
phase with disordered oxygen vacancies exists. We have used Mössbauer
spectroscopy to characterize the environment of the Fe3+ cations
in these structures. Sequences of room-temperature spectra taken from samples
with different compositions and annealed at different temperatures reveal
the presence of three distinct sites for Fe3+ in the crystal
structure, one with octahedral co-ordination, one with tetrahedral co-ordination,
and one with five co-ordinated Fe3+ (Fig. 3.3-2). We interpret
the latter as representing octahedral sites in the perovskite structure
with one adjacent oxygen vacancy. The distribution of Fe3+ between
the different sites can be determined to a first approximation from the
relative area ratios in the Mössbauer spectra. Preliminary results
show that five co-ordinated Fe3+ occurs only in the cubic disordered
phase, and that the amount varies as a function of both composition and
annealing temperature. These results combined with information from the
hyperfine parameters are being used to provide insight into the nature
of the short-range ordering of oxygen defects.
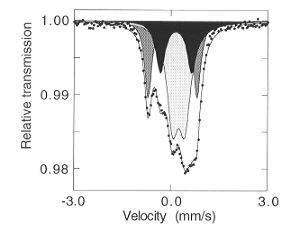 |
Fig. 3.3-2:
Room temperature Mössbauer spectrum of a CaTi0.9Fe0.1O2.95
after annealing at 900°C. The light grey, dark grey and black subspectra
correspond to octahedral Fe3+, tetrahedral Fe3+ and
pentacoordinated Fe3+, respectively.
|
Compositions with x < 0.25 invert on cooling from cubic to tetragonal to orthorhombic symmetry through a displacive phase transition. Compositions at x=0.3 and 0.4 remain cubic after rapid cooling. Using the lattice constants, a strain analysis of samples with variable Fe content annealed at high temperatures has shown that the phase transition may be first order in character. The second set of phase transitions involves ordering of the oxygen vacancies.
At high Fe contents (x>0.4) long range order in the structure, with layers of octahedra and tetrahedra, is observed corresponding to an ordered sequence of 1 tetrahedral and 3 octahedral layers for the x=0.5 composition. Different XRD patterns were observed for lower iron contents (x = 0.1-0.4) and rather low temperatures (=1100°C). The patterns show diffraction peaks corresponding to the cubic phase accompanied by several extra peaks and broad maxima. We are using high-resolution electron microscopy and electron diffraction to characterize these phases. They have a complex domain microstructure with three perpendicular orientations of planar defects. There is only one set of planar defects per domain. This complex microstructure probably reflects incompleteness of the oxygen vacancy ordering.

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page