

Since the Earth´s crust and mantle consists mostly of silicates, silica is probably the most important solute in natural hydrous fluids. Accordingly, the equilibrium solubility of the various silica polymorphs in water has been carefully studied over a wide range of temperatures. However, spectroscopic investigations of silica-bearing fluids are limited to relatively low temperatures and pressures. Accordingly, our knowledge of the dissolved silica species at high temperatures and pressures, as well as of the atomic processes occuring at the solid-fluid interface upon dissolution of silicates is rather unsatisfactory.
Here we report an investigation of silica speciation in hydrous fluids in equilibrium with solid quartz at high temperatures and pressures using an externally heated diamond anvil cell. Initial experiments were hampered by very strong fluorescence of the fluid, which made the measurement of Raman spectra impossible. This problem was found to originate from the dissolution of traces of rhenium used as gasket material. Upon replacing the rhenium by iridium, the fluorescence disappeared completely and Raman spectra could be measured at all pressures and temperatures accessible by the cell. Raman spectra were recorded with a Spex 1877 triplemate spectrometer equipped with a Photometrics CCD detector cooled by liquid N2. The 514 nm line of a coherent argon laser was used as excitation source in 155°C scattering geometry. Measurements were made in the 350 - 1050 cm-1 spectral range to locate the Si-O vibrational modes, and in the 2800 - 3800 cm-1 spectral range in order to study the O-H stretching modes. The accumulation times ranged from 120 to 2000 seconds per spectral window.
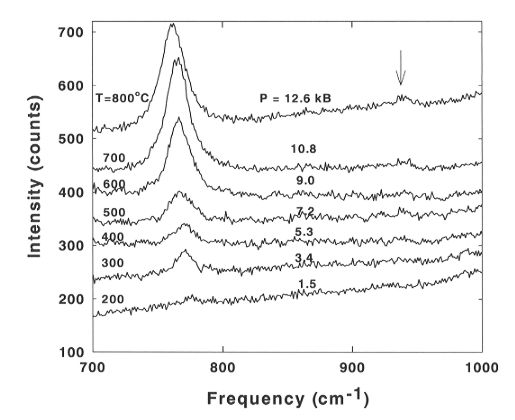
Some representative Raman spectra are shown in Fig. 3.5-1. With increasing
temperature and pressure the peak at about 770 cm-1 increases
in intensity. This peak is due to the symmetric stretching vibration of
the H4SiO4 monomer (Qo species). Measurements
of the Qo peak upon heating and cooling with different delay
times show a reproducibility of the peak position to less or equal 1 cm-1
and of the integral intensity to less or equal 25%. Measurements on deuterated
fluids showed an isotopic shift of the Q0 band of about 1.008
± 0.001 which clearly demonstrates that it originates from a hydrous
species. Measurement on fluids with different bulk densities revealed the
effects of temperature and pressure on the frequency of the Q0
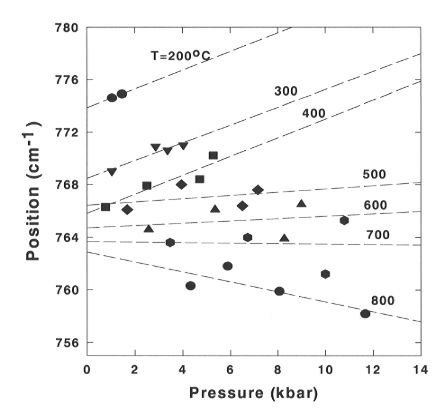
peak (Fig. 3.5-2). The position of the Q0 peak varies linearly
with pressure for all temperatures studied. With increasing temperature,
the slope of the curves starts to decrease, and above 600°C it becomes
negative which indicates a significant anharmonicity and softening of the
Si-(OH) bond, which counteracts the effect of increasing pressure.
 |
|
Fig. 3.5-1: Raman spectra of hydrous fluid saturated with quartz,
measured under isochoric conditions in a diamond anvil cell. Bulk density
of water was 0.95 g/cm3. The arrow points to a weak peak only
detectable at the highest pressures and temperatures.
|
The intensity of the Q0 peak increases strongly with increasing pressure and temperature which reflects the increasing solubility of quartz. At very high pressures and temperatures a new peak starts to develop at about 940 cm-1 (marked by an arrow on Fig. 3.5-1) which we assign to a polymerized species (e.g. H6Si2O7). The Raman spectra of the quartz crystal in contact with the fluid also show the appearance of a new peak at about 900 cm-1 which indicates the formation of Si-OH groups on the surface as an intial step of the dissolution. Additional studies are in progress to determine the equilibrium constant for the reaction 2 H4SiO4 = H2O + H6Si2O7, the P-T dependence of the silica speciation and the effects of pressure and temperature on the hydrogen bonding in liquid water in the presence of dissolved silica.
 |
|
|

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page