

It is now well established that nominally anhydrous minerals in the mantle can dissolve substantial amounts of OH as point defects. However, more information is needed about the partitioning of water among all of these phases in order to understand how water is distributed in the mantle and to which extent water is recycled by subduction. We conducted multianvil experiments in the system MgSiO3 - H2O with a small excess of silica at pressures and temperatures representative for the transition zone and lower mantle. The recovered samples were characterized by Raman spectroscopy and the water contents were measured on optically clear crystals using a micro-FTIR spectrometer. The extinction coefficients were derived from the calibration by Paterson. The phases synthesized were clinoenstatite, majorite, MgSiO3-ilmenite, MgSiO3-perovskite, and ringwoodite. All these minerals coexisted with stishovite. In runs conducted above 1200°C, a melt was present as well. The results of the IR analysis show that among all nominally anhydrous magnesium silicate phases (1) MgSiO3-perovskite is the only phase which does not dissolve any detectable amount of water, and (2) the minerals having Mg2SiO4 composition, namely ringwoodite and wadsleyite, can incorporate up to 2-3 wt% H2O, whereas the polymorphs of MgSiO3 only dissolve up to 2500 ppm of water under the same conditions.
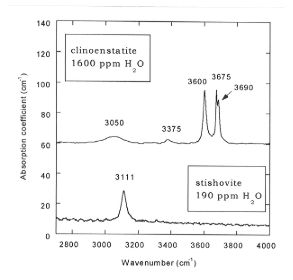
The FTIR spectra of clinoenstatite consist of three strong and sharp
bands at low frequency and two weak and broad bands at higher frequency
as shown in Fig. 3.5-3. The partition coefficient of water between stishovite
and clinoenstatite (Dclinoenst/stish) was found to be less than
20.
 |
|
|
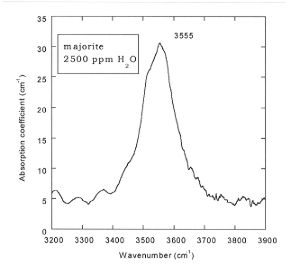
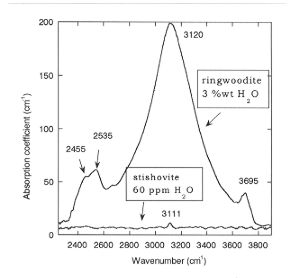
We previously found a similar value for the partitioning between stishovite and ilmenite, whereas Dmaj/stish is about 250. Fig. 3.5-4 shows the FTIR spectra of majorite which consists of a single band at 3555 cm-1. The FTIR spectra of ringwoodite (Fig. 3.5-5) displays a strong absorbance band at 3120 cm-1 and weaker bands at 3695, 2535 and 2455 cm-1, which are consistent with ~ 3 wt% H2O. The partition coefficient of water between ilmenite and ringwoodite coexisting in the same sample Dringw/ilm is 20 whereas Dringw/stish is 500.
These results show that pure MgSiO3 perovskite is not likely
to store significant amounts of water. Nevertheless, the solubility of
water in perovskite could increase in the presence of chemical impurities,
in which case deep subduction could recycle water into the lower mantle.
 |
|
|
 |
|
|

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page