

Carbonatite magmas only rarely erupt at the Earth´s surface, but they are believed to play a major role in mantle metasomatism. According to field evidence, these melts are probably very rich in water and water is also partially responsible for some unusual properties of these melts, such as their low melting point and extreme mobility. There are, however, no experimental data on the solubility of water in carbonatite magmas. This is not surprising, since carbonatites cannot be quenched to glasses and therefore, conventional methods for measuring water solubility cannot be applied to these melts. In order to solve this problem, a new experimental method was developed which for the first time allowed the solubility of water in liquid carbonatite to be measured.
About 70 mg of carbonate (40 wt% CaCO3, 40 wt% Na2CO3
and 20 wt% MgCO3) were sealed into a platinum capsule. This
capsule was run above the liquidus in a rapid-quench autoclave at 1 kbar
and 900°C. Upon quench, the melt solidified to a massive block of carbonate
inside the capsule. A tiny pinhole was drilled into the uppermost end of
the capsule, which was then sealed in a larger platinum capsule together
with about 50 mg of water. In the following experiment, the double capsule
was always kept vertical, with the pinhole of the inner capsule pointing
upward. In this way, the molten carbonatite could not flow out of the inner
platinum container. A rapid-quench autoclave was preheated to 1 kbar and
900°C with the capsule being in the cold part of the vessel. Once the
final temperature was reached, the capsule was elevated into the hot spot
using an external magnet actuating a magnetic filler rod inside the bomb.
After a few hours, the charge was quenched to room temperature within 1
- 2 seconds and removed from the vessel. The outer capsule was opened and
removed, the inner capsule mechanically cleaned and weighed. Visual inspection
showed that the capsule was usually filled with massive quenched carbonate
up to the pinhole. Small quantities of carbonate often were extruded from
the inner capsule. To avoid water loss prior to the measurement, the inner
capsule was immediately sealed into a new platinum container. The sample
was then placed in a Karl-Fischer-titration apparatus and the outer capsule
pierced immediately before the measurement. Upon controlled heating, water
was released from the quenched carbonatite and measured by coulometry.
Most of the water was lost between 200 and 300°C, which suggests the
presence of hydroxyl groups in the sample.
 |
|
|
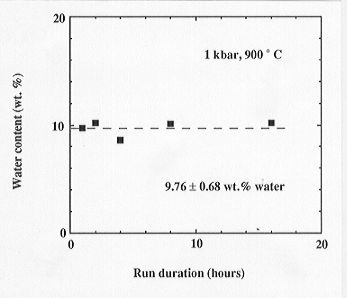
The first data on water solubility in a carbonatite melt ever measured are shown in Fig. 3.5-13. Water solubility is 9.76 ± 0.68 wt% at 1 kbar and 900°C, much higher than for most silicate melts. These data are consistent with the signs of massive hydrothermal alteration usually observed in the vicinity of carbonatite bodies. They also suggest that complete miscibility between carbonatite melt and water may already be reached in the deeper crust.

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page