

The transport properties of silicate melts within the Earth strongly influence processes such as magma ascent, crystal fractionation and convection within magma chambers. Of particular interest in this study is the pressure dependence of viscosity for compositions relevant to those of the Earth's deep interior. This is an important factor in modeling convection and crystal fractionation in planetary sized magma oceans that are thought to have extended to depths greater than 1000 km in the Earth. It is a widely accepted theory that magma oceans played an important role in the early differentiation of the Earth and other planets and in the formation of the moon. The present geochemical and geophysical structure of the Earth may be better explained if, for example, rigorous convection in a magma ocean forced crystals to remain in suspension allowing full equilibration between crystals and melt before solidification. Viscosity is a critical parameter in this model and, in previous work, has been assumed to be unaffected by pressure.
In order to understand better the pressure dependence on the transport properties of melts relevant to the Earth's deep interior, the self-diffusion of oxygen and silicon were measured in diopside melt at 2000°C and up to 8 GPa. A rhenium capsule containing diopside powder enriched in 18O and 30Si (lower half) and unenriched diopside powder (upper half) was compressed in a multianvil apparatus. Run times were generally short (ca. 2 min.) because of the very high temperature and short length required of the specimen. The resulting diffusion profiles were measured by secondary ionisation mass spectrometry (SIMS) at the University of Edinburgh, Scotland.
By measuring the diffusion coefficients of the species that control viscous flow (silicon and oxygen), it is possible to use the Eyring relation to calculate viscosity. This approach to measuring the viscosity of polymerised melts (NBO/T = 0-0.5) at high-pressure (up to 15 GPa) has proven to be a successful alternative to the more traditional falling sphere method. However, it has been suggested that the unit of viscous flow and therefore the diffusive jump distance might be larger for more depolymerized melts.
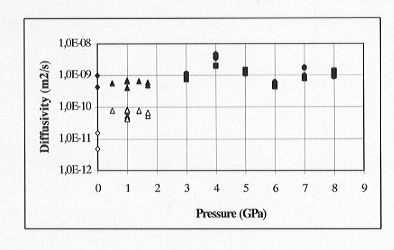
Our preliminary results (Fig. 3.6-12) indicate that oxygen and
silicon diffusion are identical within experimental error (log DSi
= log DO = -9 ± 0.5 (m2 s-1)) and
that there is no pressure effect on either oxygen or silicon self-diffusion
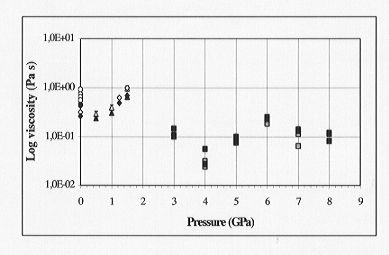
at 2000°C in the pressure range 3-8 GPa. The calculated viscosity at
this temperature using the Eyring equation is 0.1 Pa s (Fig. 3.6-13). This
is slightly lower than previous falling sphere measurements of diopside
(up to 1.5 GPa). However these may overestimate viscosities due to the
assumption that the sphere falls (or rises) in the melt as soon as the
desired experimental temperature is reached. Efforts to conduct in-situ
falling sphere viscosity measurements using the electro-detection method
(described below by Secco and Poe) and synchrotron radiation are also underway.
 |
|
Fig. 3.6-12: Self-diffusion of silicon and oxygen in diopside
melt up to 8 GPa (2000oC, 120 s). Filled circles = oxygen (this
study), filled squares = silicon (this study), open triangles = oxygen
and silicon 1650oC (Shimizu and Kushiro, 1984; Shimizu and Kushiro,
1991), open diamonds = oxygen 1400-1500oC (Dunn, 1982), filled
triangles and diamonds = above literature data temperature corrected to
2273K.
|
 |
|
Fig. 3.6-13: Viscosity of diopside melt up to 8 GPa (2273 K):
Literature data from falling sphere measurements (up to 2 GPa) and values
calculated from diffusion coefficients of silicon and oxygen (this study)
using the Eyring equation. Grey box = oxygen (this study), filled square
= silicon (this study), open triangles = 1873 K (Brearley et al. 1986),
open diamonds = 1913 K (Scarfe et al. 1979), open circles = 1750 K (Scarfe
et al. 1987), filled triangles, diamonds and circles = above literature
data temperature corrected to 2273K.
|

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page