

Recent work in Paris has shown that temperature gradients in laser-heated
diamond anvil cell experiments can have large effects on oxygen fugacity
conditions, and therefore potentially large effects on physical properties
such as electrical conductivity. To quantify the amount of Fe3+
in quenched phases synthesised by laser heating, we used the Mössbauer
milliprobe (Annual Report, 1993), which enables different areas of the
sample to be studied (Fig. 3.2-6).
 |
Fig. 3.2-6: 57Fe0.15Mg0.85SiO3 orthopyroxene after laser heating in the diamond anvil cell. The horizontal stripe shows the area that was heated. Mössbauer spectra were taken from three areas: (1) transparent area in the middle (perovskite phase); (2) dark area at the edges (heated, not transformed); and (3) white area (unheated). The diameter of the spots studied with Mössbauer spectroscopy (indicated by circles) is 100 µm. |
We found that Fe3+ only occurs in the perovskite
phase; no evidence for Fe3+ was found in either the
unheated or heated areas of orthopyroxene. In experiments with (Mg,Fe)O,
we found that samples heated in the diamond anvil cell become transparent
and contain significantly less Fe3+ than the starting
material, consistent with results from multianvil press experiments (Annual
Report, 1995). Experiments were also performed to compare results from
orthopyroxene loaded in air and under argon. Mössbauer spectra recorded
at the centres of heated spots showed a significant difference: samples
loaded in air gave Fe3+/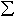 Fe values of 5-7% in 57Fe0.15Mg0.85SiO3
perovskite; whereas samples loaded in argon gave values of 2-3% in the
same phase. In addition, there was a magnetic phase present in the samples
loaded in argon whose parameters corresponded to metallic iron. No magnetic
phase was detected in the samples loaded in air.
Fe values of 5-7% in 57Fe0.15Mg0.85SiO3
perovskite; whereas samples loaded in argon gave values of 2-3% in the
same phase. In addition, there was a magnetic phase present in the samples
loaded in argon whose parameters corresponded to metallic iron. No magnetic
phase was detected in the samples loaded in air.

Tel: +49-(0) 921 55 3700 / 3766, Fax: +49-(0) 921 55 3769, E-mail: bayerisches.geoinstitut(at)uni-bayreuth.de
 Previous page
Previous page